Sustainability has become a top priority for businesses and now shapes how they are evaluated and trusted. Companies in every industry are judged not only on their results and profits, but also on how they operate. In life sciences, using sustainable practices in clinical trials is an important factor when sponsors and CROs pick their partners and service providers. ESG, which stands for Environmental, Social, and Governance, provides organizations with a framework for measuring these efforts and setting clear standards for accountability.
ESG in Clinical Research Is Now an Expectation, Not an Option
Sustainability has become a strategic necessity that belongs at the center of every organization’s operations and long-term planning, driven by both ethical responsibility and financial performance. Today’s workforce increasingly seeks purpose-driven employers that demonstrate genuine environmental commitment, with a strong majority of job seekers viewing sustainable companies as more attractive places to work.
Consumers are also aligning their spending habits with values, showing a clear willingness to support and even pay more for environmentally responsible brands. At the same time, global governments, investors, employees, and customers are calling for greater corporate accountability, particularly in addressing climate change.
While ESG may be another acronym in an industry full of them, it carries real weight. It influences investor confidence, talent attraction and retention, supplier selection, and long-term success. Increasingly, it also shapes how sponsors and CROs evaluate their partners.
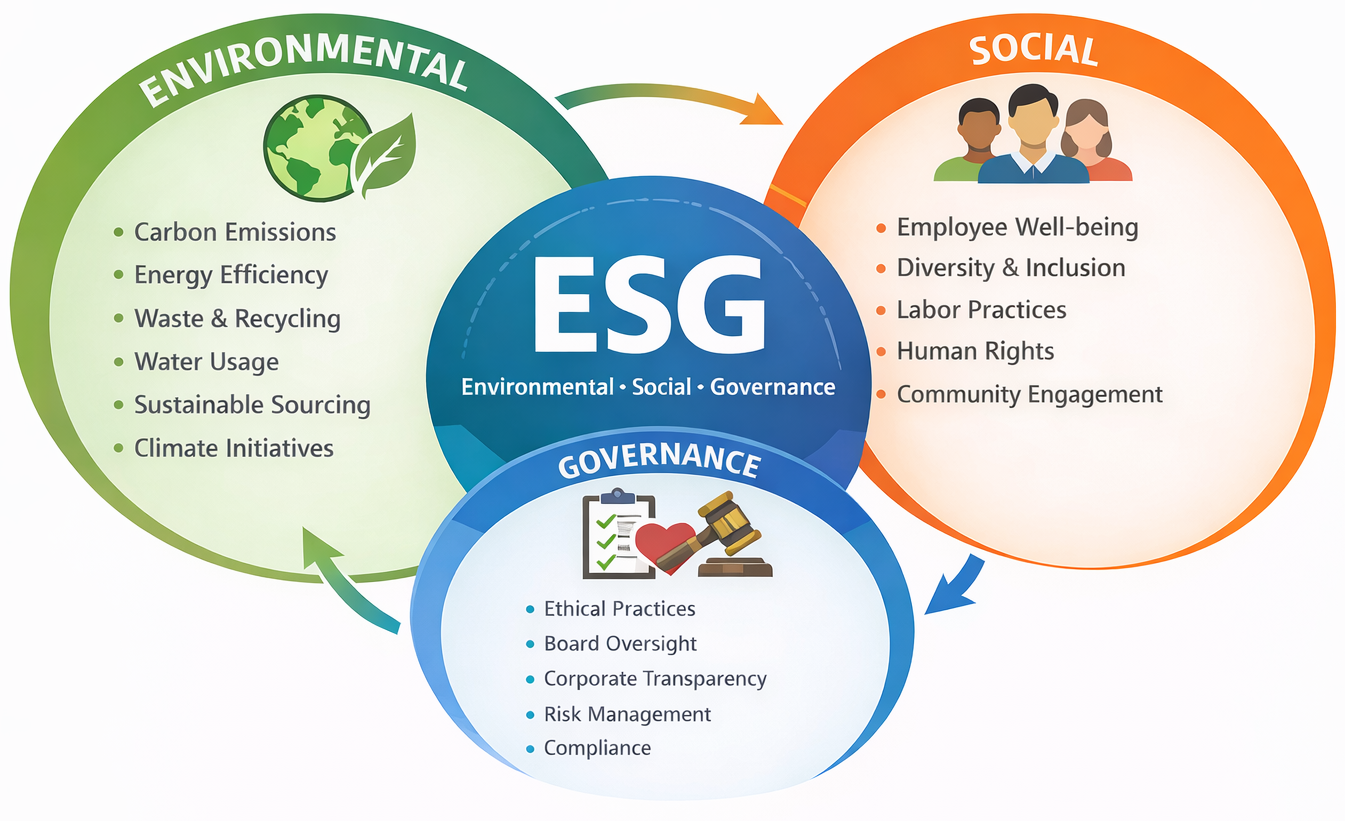
What ESG Encompasses
ESG standards evaluate companies across three core pillars:
Environmental – Impact on the Planet
- Greenhouse gas emissions and pollution control
- Energy usage and efficiency
- Waste reduction and recycling
- Sustainable sourcing practices
- Environmental policies and measurable goals
Social – Impact on People
- Diversity, equity, and inclusion initiatives
- Employee health and safety
- Human rights protections
- Fair labor practices and wages
- Community engagement and charitable efforts
Governance – How a Company Operates
- Ethical business conduct
- Leadership accountability
- Executive oversight and transparency
- Risk mitigation practices
- Compliance frameworks
By prioritizing ESG criteria, companies can mitigate risks, attract top talent, and ensure long-term profitability. In clinical research, sponsors and CROs are now incorporating ESG factors into vendor qualification, RFPs, and capability assessments. Even when sustainability isn’t the primary focus, teams often integrate it into operational due diligence.
Putting ESG to the Test
Imperial takes its role as a corporate citizen seriously and has long maintained policies and procedures aligned with these principles. In recent years, we have worked intentionally to formalize, document, and strengthen them, ensuring they are measurable, transparent, and aligned with the expectations of the global sponsors and CROs we support.
To benchmark our efforts, Imperial again participated in an independent evaluation through EcoVadis, a Paris-based global sustainability ratings provider widely recognized for its rigorous methodology.
EcoVadis Bronze Medal Recognition for 2026

We are proud to share that Imperial has been awarded the EcoVadis Bronze Medal for 2026.
This recognition places Imperial among the top 35% of companies evaluated globally by EcoVadis in our category. It reflects measurable progress across key sustainability performance indicators. The rating also shows that Imperial’s performance aligns with top-tier sustainability practices worldwide.
The EcoVadis assessment evaluates 21 sustainability criteria across core themes:
- Environment: Reducing energy consumption, demonstrating water stewardship, and implementing environmental policies
- Labor & Human Rights: Employee health and safety, diversity and inclusion benchmarks, fair labor practices, and human rights due diligence processes
- Ethics: Anti-corruption training, data protection practices, whistleblower mechanisms, and formal compliance policies
- Sustainable Procurement: Supplier ESG screening rates, adopting a supplier code of conduct, supply chain risk assessments, and responsible sourcing documentation
The assessment process is comprehensive and includes extensive documentation review, policy validation, and evidence-based scoring. It also provides invaluable insights in the form of a detailed roadmap for continuous improvement, identifying areas where stronger data tracking, formalized policies, or expanded supplier engagement can enhance performance in the coming year.
Why Independent Benchmarking of ESG Matters in Clinical Research
Sustainability claims without verification carry little weight. Independent benchmarking provides credibility, accountability, and comparability.
EcoVadis also functions as a secure information-sharing platform. For example, clients and prospective partners can access Imperial’s scorecard directly, reducing RFIs and increasing transparency.
In life sciences, especially, ESG performance increasingly influences supplier selection. Large pharmaceutical companies and global CROs expect measurable sustainability commitments from their partners.
ESG in Action at Imperial
Sometimes, sustainability shows up in small but meaningful decisions. At industry conferences, for example, we evaluated the traditional promotional giveaways — often single-use plastic items — and chose a more responsible alternative.
Instead of disposable gadgets, we distribute “second life” giveaways, such as plantable seed pencils and bamboo eco-friendly planter kits. The response has been overwhelmingly positive. More importantly, it aligns our brand presence with our values.

Imperial is committed to enhancing our environmental stewardship, social responsibility, and governance practices while continuing to refine our processes to achieve measurable improvements. Working together, we can make a truly lasting impact.
Resources
- EcoVadis https://ecovadis.com/
- Sustainability in Business: What It Is and Why It Matters https://www.seattleu.edu/business/online/albers/blog/what-is-sustainability-in-business
- The Sustainability Puzzle: What Do Consumers Really Want? https://www.bain.com/insights/what-do-consumers-really-want-ceo-sustainability-guide-2024/
- What is ESG and why does it matter for businesses? https://fsc.org/en/blog/what-is-esg
- What is sustainability in business? https://www.ibm.com/think/topics/business-sustainability

